Biologics candidates often fail late in development due to sequence liabilities, aggregation, or unwanted immune activation. These setbacks can result in lost investments, missed partnership opportunities, impact on reputation and prolonged timelines.
Early de-risking protects you and your molecule. It is designed to help answer the most vital questions about your biologic, giving you decision‑ready, IND‑relevant data, so you can prioritize your best candidates, shape later CMC and enter into investor conversations with confidence.

Fewer costly surprises
Identify critical risks earlier.
Stronger IND story
Clear rationale, clear next steps.

Better investor conversations
Sharper, differentiated data package.
What’s in the Early De-Risking Package?
1. In silico developability assessment
Identify sequence liabilities such as post translational modifications, aggregation, and immunogenicity to support candidate selection.
2. Stable pool protein expression
Assess your molecule in a commercially-relevant GS Expression System® to
de-risk scale up and product development.
3. In vitro peripheral blood mononuclear cells (PBMC) activation assay for cytokine storm risk
Screen for cytokine storm risk, a critical patient‑safety concern.
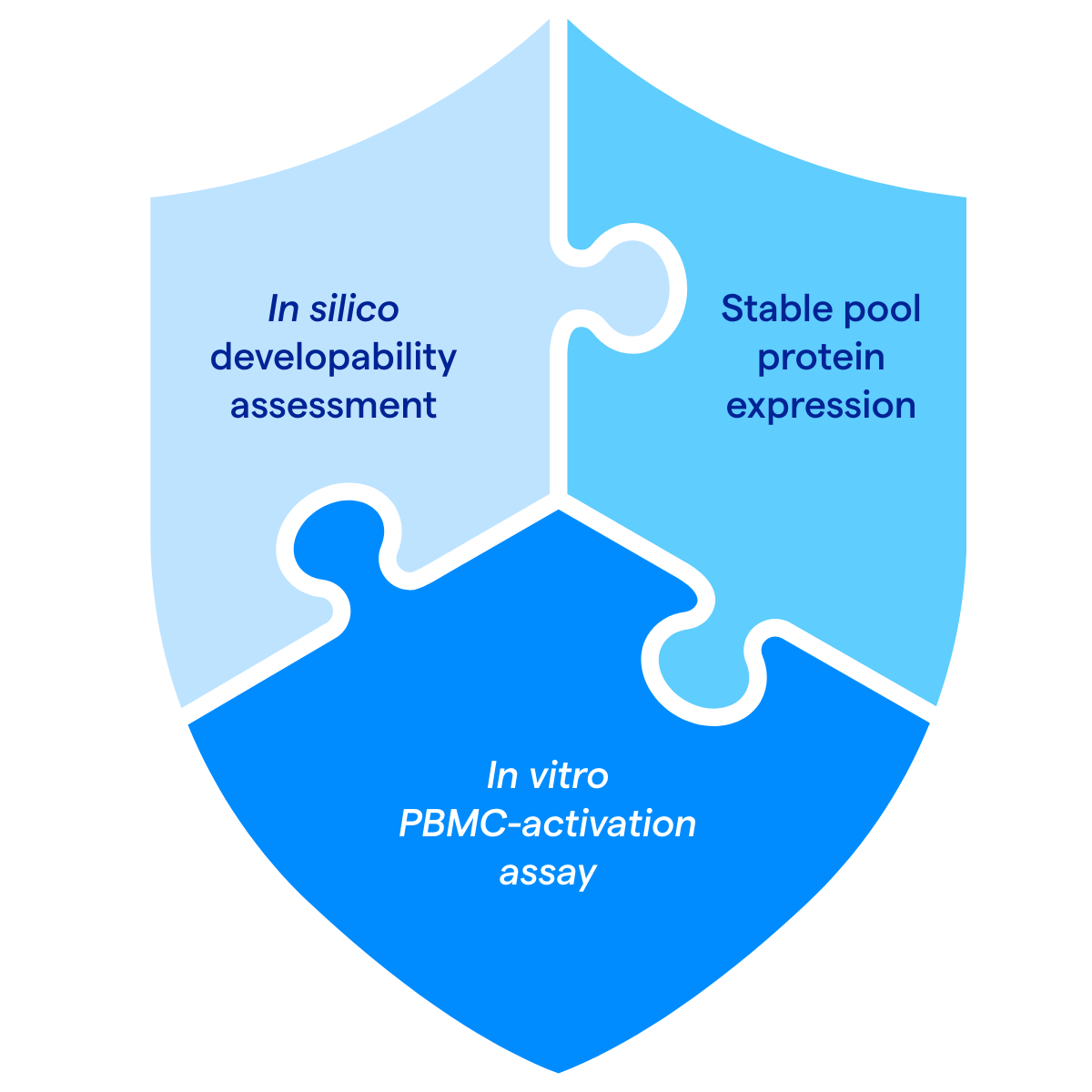
An integrated 3‑step package to assess safety and manufacturability
in as little as 11 weeks
What you give us
Your sequence
What we do
In silico analysis of your sequence for developability
Expressing your biologic in our GS System® to de-risk scale up and your manufacturing journey
In vitro screening for immunosafety risk with cytokine storm risk assay
What you get
Consolidated report with IND‑relevant, decision‑ready data.
- Outline of the main risks highlighted.
- Our overall assessment.
- Risk mitigation strategies (where appropriate).
Ready to de-risk your molecule?

A comprehensive and robust de-risking package at lower cost.

Integrated product assessment across two key developability questions: Is it safe? Can it be manufactured?

An optimized timeline from receipt of sequence(s) to IND-relevant, decision-ready data.
De-risk the science. Strengthen the story. Accelerate the capital.
Complete the form to get started


